Intoduction
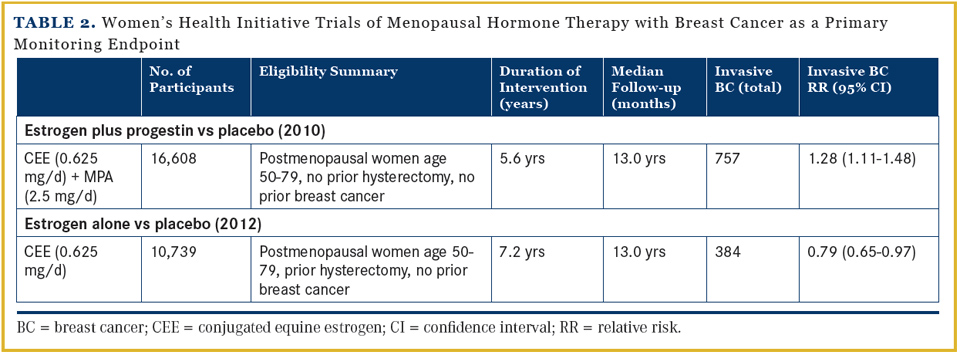
Both tamoxifen and raloxifene have FDA approval for breast cancer risk reduction. These recommendations are endorsed by an American Society of Clinical Oncology (ASCO) guideline,1 by the National Comprehensive Cancer Center Network (NCCN) guideline,2 and, importantly, by the US Preventive Services Task Force (USPSTF).3 Despite these recommendations, uptake of selective estrogen receptor modulators (SERMs) for breast cancer chemoprevention in clinical practice has been extremely limited.4 In addition, updated, long-term follow-up of two of these chemoprevention trials raises questions regarding the role of tamoxifen in this setting. Updated analyses from the Women’s Health Initiative (WHI) randomized hormone therapy trial (Table 2) have informed understanding of the relationships between exogenous estrogen and exogenous progestin and breast cancer risk, with relevance for breast cancer prevention.
SERMs and Breast Cancer
In IBIS-I,6 7154 women were randomized to either tamoxifen or placebo for 5 years in a primary breast cancer prevention trial. Study findings were recently updated with 16 years’ follow-up.17 A reduced breast cancer incidence persisted throughout follow-up (214 vs 289 cases; hazard ratio [HR], 0.73; 95% CI, 0.61-0.84; P <.0001). However, there were more deaths in the tamoxifen group (187 [5.1%]) vs 166 deaths [4.6%], respectively; odds ratio [OR], 1.10; 95% CI, 0.88-1.37)17 (Table 1). In an accompanying commentary, Chlebowski questioned the greater mortality in the tamoxifen group and suggested that although the findings could reflect the play of chance in a small sample, less favorable alternative explanations include tamoxifen only decreasing breast cancers with favorable prognosis or tamoxifen increasing breast cancers with unfavorable prognosis.18
The NSABP STAR trial for breast cancer prevention also recently was updated.19 Intervention was originally for 5 years with median 9.7 years follow-up. Tamoxifen reduced invasive breast cancers compared with raloxifene (375 vs 453 cases; relative risk [RR], 1.19; P = .01).19 However, there were 5 more deaths from breast cancer in the tamoxifen group and more deaths from all causes as well (413 vs 364 deaths, respectively), a finding of borderline statistical significance (RR, 0.87; 95% CI, 0.75-1.00). Taken together, the findings from IBIS-I and the STAR trials challenge the primary role of tamoxifen for chemoprevention in postmenopausal women. These findings are difficult to understand since in an adjuvant setting, after 15 years follow-up, tamoxifen not only reduced breast cancer recurrence risk but improved overall survival, as well.20
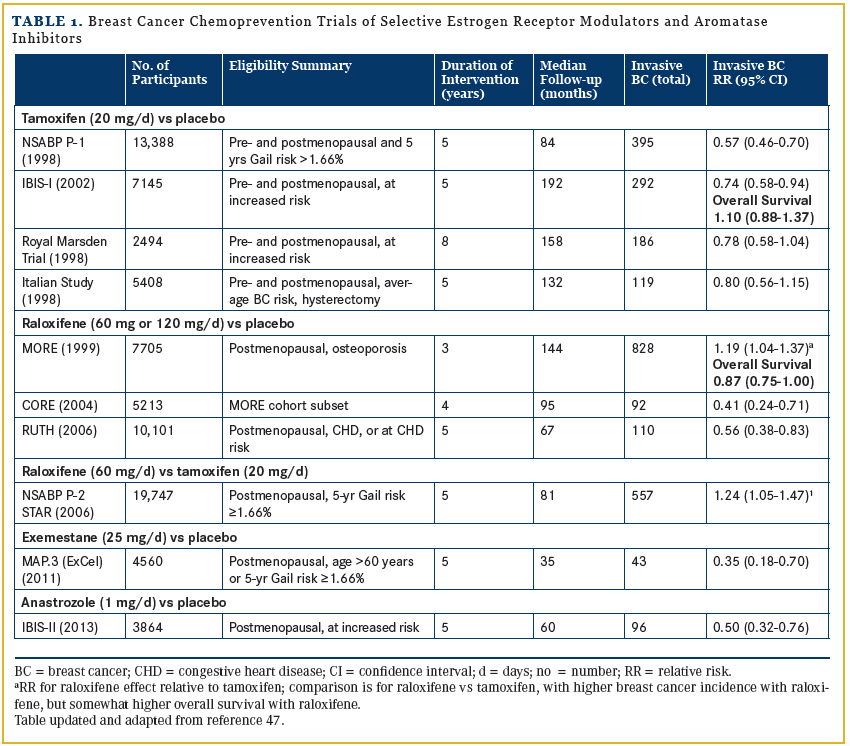
Aromatase Inhibitors and Breast Cancer Risk
Two full-scale trials have evaluated aromatase inhibitors (AIs) for breast cancer prevention based on their efficacy in reducing contralateral breast cancer in adjuvant breast cancer trials.21 In the Mammary Prevention 3 (MAP.3) trial,22 postmenopausal women were eligible based on age alone (≥60 years) or increased breast cancer risk. A total of 4560 women were randomized to exemestane 25 mg or placebo. Exemestane reduced breast cancer risk by 65% (HR, 0.35; 95% CI, 0.18-0.70; P = .002).22 Similarly, in the International Breast Cancer Intervention Study II (IBIS-II),23 a randomized, placebo-controlled breast cancer prevention trial with 3864 postmenopausal women, the AI anastrozole reduced breast cancer risk by 53% (HR, 0.47; 95% CI, 0.32-0.68; P <.00001). In addition, there were significantly fewer other cancers in the anastrozole group, including skin and colorectal cancers (RR, 0.53; 95% CI, 0.28-0.99).In adjuvant breast cancer trial reports, compared with tamoxifen, AIs increased fractures and substantially increased musculoskeletal complaints. In the placebo-controlled breast cancer prevention trials, a different toxicity profile emerges.22,23
In the MAP.3 trial,24 no increase in fractures was seen, the frequency of musculoskeletal side effects was lower than in adjuvant trials, and global quality of life did not differ between randomization groups. Similar findings were reported in the IBIS-II study,23 with no increase in fractures with anastrozole use and modest differences in musculoskeletal complaints between anastrozole and placebo users (7% vs 5%, respectively).
With respect to fracture-risk differences seen in the adjuvant versus prevention setting, the adjuvant trials were conducted when there was limited understanding of bone health. Subsequently, bone mineral density (BMD) monitoring and bone-targeted therapies have come into standard clinical practice. In both the MAP.3 and the IBIS-II trials, serial BMD monitoring was not a protocol requirement and bisphosphonate use was not protocol-defined. Nonetheless, in these trials, the 15% to 25% use of bisphosphonates reflected current clinical practice largely directly by primary care physicians.22,23 Thus, current AI use would not be expected to increase fracture risk in women receiving current medical management.
The difference in musculoskeletal symptoms for AI use in the adjuvant setting compared with the primary prevention setting could reflect differences in characteristics of participants. Woman in prevention trials were older; had not received therapy likely to exacerbate joint symptoms, such as chemotherapy and radiation therapy; and many were taking medications for prevention, and thus might expect few additional problems when adding another medication.25
Menopausal Hormone Therapy and Breast Cancer Risk
When the 2 WHI hormone therapy clinical trials, separately evaluating estrogen plus progestin in women with a uterus and estrogen alone in women with prior hysterectomy, were initiated, an increase in breast cancer was anticipated in both—but the cancers were anticipated to have favorable characteristics. The surprising results of these 2 studies have fundamentally changed understanding of the relationship of exogenous estrogen and progestin to breast cancer.26,27The WHI estrogen-plus-progestin trial was stopped when more harm than benefit emerged for combined hormone therapy use.28 Estrogen plus progestin significantly increased breast cancer incidence,29 interfered with mammographic cancer detection,29,30 and significantly increased breast cancer mortality.31 In contrast, the WHI trial evaluating estrogen alone in postmenopausal women with prior hysterectomy found that estrogen alone significantly decreased breast cancer incidence32,33 and significantly decreased deaths from breast cancer.33
When the findings for estrogen-plus-progestin use were reported in 2003, a rapid decrease in hormone therapy use occurred in the United States and around the world,34,35 which was associated with the first decrease in breast cancer incidence in the United States in over 20 years.36,37 While subsequent reports generally supported the original findings,35 there were questions about whether the rapid drop in breast cancer was biologically feasible or was related to a reduction in mammography use.
In the WHI clinical trial, all participants were instructed to stop their study medication when the intervention ended with 98% compliance. In addition, mammography frequency was similar immediately before and after the intervention ended in both randomization groups. The end of the intervention was followed by a rapid and statistically significant reduction in breast cancer incidence.38 Taken together, these findings supported the prior proposed hypothesis. The immediate reduction in breast cancer incidence was felt to reflect the impact on preclinical but already established breast cancers from a sudden change in hormone environment, similar to an oophorectomy or AI effect in women with established breast cancer.38
After 5.6 years of estrogen-plus-progestin use, with an additional 8 years of postintervention follow-up, a persistent elevation in breast cancer risk of about 24% developed.39 A year-to-year analysis comparing the intervention period to the post- intervention period identified a complex pattern. There was a significant increase in year-to-year risk during intervention, a sudden decrease in risk post-intervention for about 2 years, followed by sustained increase in breast cancer incidence afterwards (Figure 1).27
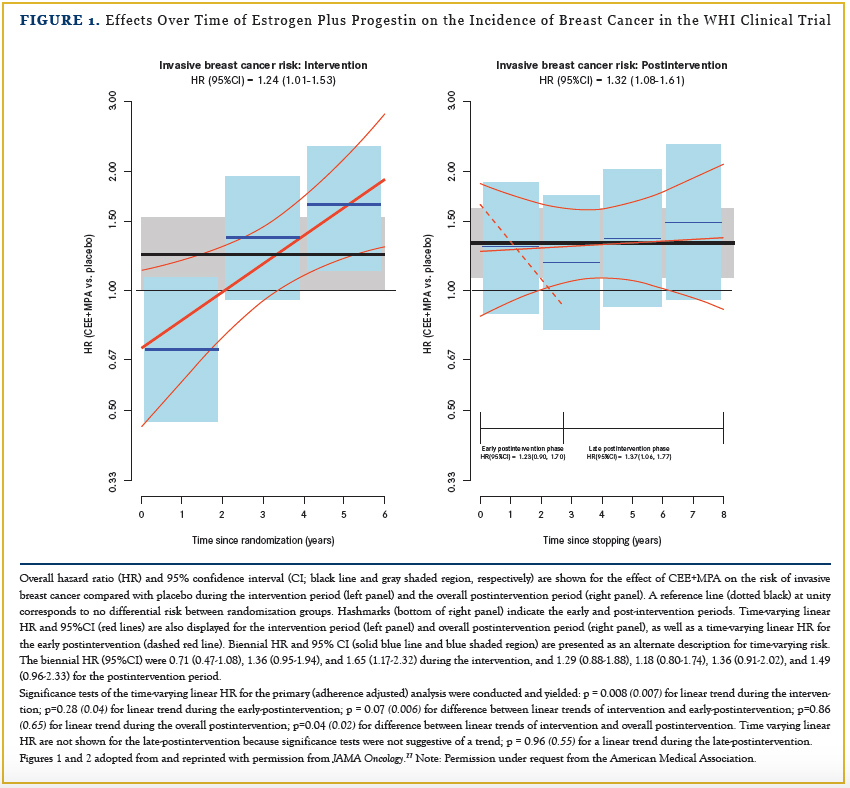
In an editorial, Joshi and colleagues40 posed a biologically plausible explanation for these findings. As previously suggested,38 the initial post-intervention decrease was felt to be related to a reduction in estrogen exposure and resulting inhibition of growth of hormone receptor (HR)-positive preclinical breast cancers. However, the authors postulated that progestin, demonstrated to stimulate breast mammary stem cells in preclinical studies, then results in an excess of stem cells responsible for the long-term, sustained increase in breast cancer risk. As a result, the long-term risk of breast cancer for estrogen plus progesterone for about 5 years of use is substantially greater than previously thought.
The findings regarding breast cancer for estrogen-alone use in women with prior hysterectomy were opposite those in the combined-hormone-therapy trial. Estrogen-alone use significantly decreased breast cancer incidence and significantly decreased deaths from breast cancer.33 When examined for year-to-year influence on breast cancer incidence during the intervention and post-intervention period, a lower breast cancer incidence was seen throughout the intervention, which persisted for about 4 years post intervention (Figure 2).27
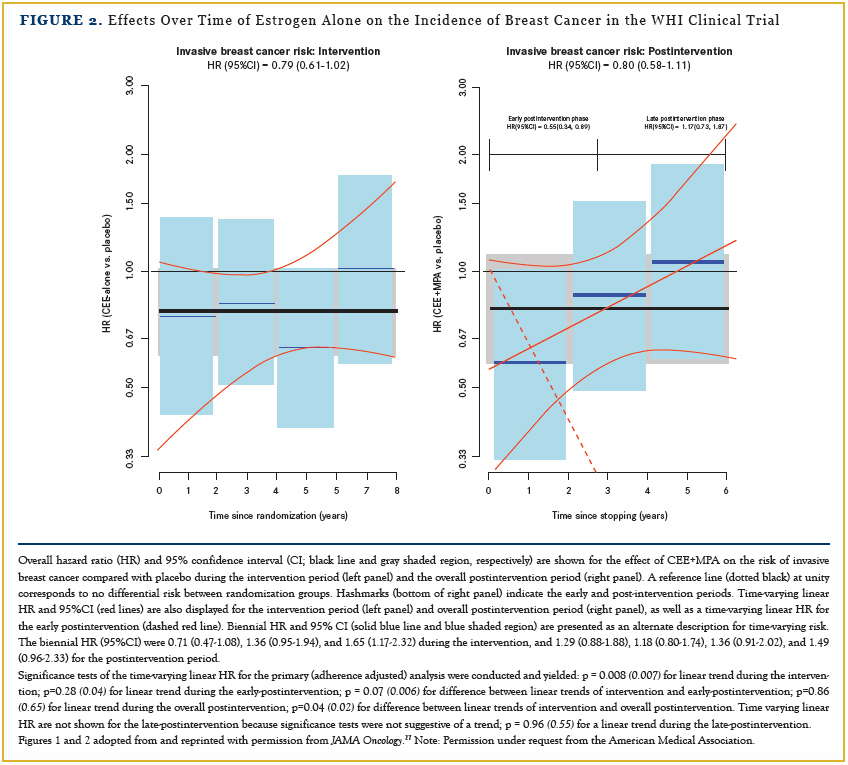
The effect appeared to be more pronounced in black women, particularly those with more than the median percentage of African ancestry (>80%) in whom a 68% reduction in breast cancer incidence was seen.41 Thus, the favorable breast cancer effect of estrogen-alone use in black women has identified a potentialintervention strategy for addressing the disparity in breast cancer mortality risk seen in black compared with white women in the United States.
In the chemoprevention trials, estrogen reduction with AIs resulted in a reduction in breast cancer incidence.23,24 However, in the WHI trial, estrogen addition with conjugated equine estrogen also resulted in a reduction in breast cancer incidence.27,33 A likely explanation for this apparent paradox has been put forward by Jordan and colleagues.50 Estrogen typically simulates mammary epithelium and inhibits apoptosis to prepare the breast for milk production. However, after a period of estrogen deprivation, gene-expression profile change results in estrogen functioning as an apoptosis stimulant.42 These time-dependent, exposure level–dependent, complex interactions have practical clinical implications. Ellis and colleagues42 provided proof-of-principle in a study where postmenopausal women with HR-positive, advanced breast cancer refractory to AI use had some demonstrated activity to relatively low doses of estradiol (2 mg/ day). Taken together, these findings suggest that breast cancers that are HR-positive can survive only in an environment with a relatively narrow estrogen range.
Breast cancer prevention is commonly felt to be quite distinct from breast cancer therapy. However, a recent report by Santen and colleagues43 calls that concept into question. A tumor-growth kinetic model, based on preclinical and clinical findings of tumor-doubling time, size detection threshold, and tumor prevalence based on autopsy series was used to model the percentage of breast cancers in the WHI trial evaluating estrogen plus progestin, where age and eligibility criteria are similar to those in most breast cancer chemoprevention trials. Findings from the model analysis indicated that 94% of cancers detected were already established but preclinical, whereas only 6% were de novo tumors. Thus, it is likely that breast cancer chemoprevention in prevention trials can be, at least in part, therapy of preclinical breast cancer.
Menopausal Hormone Therapy and Breast Cancer in Observational Studies
The findings from the WHI hormone therapy randomized trials on breast cancer incidence and outcome differ in many ways from many observational studies of the same issue. For example, in the Million Women’s Study, although no increase in breast cancer is seen with estrogen-alone use in women starting use in their fifth decade, no decrease in incidence is seen.48 The discussion of bases of such differences is beyond the scope of the present report, but the issue has been addressed elsewhere.26,27,47
Breast Cancer Chemoprevention and Linear Relationships
Based on preclinical and emerging observational study evidence,44-46 metformin is currently undergoing trial as an addition to standard adjuvant breast cancer therapy in a randomized trial with over 3500 randomized patients. Because metformin has a well-established safety profile, if this adjuvant trial is positive with an impact on contralateral breast cancer, one could reasonably propose metformin for breast cancer prevention use.
Conclusions
Affiliation: Rowan T Chlebowski, MD, PhD; Radhika Prabhaker, MD; and Tomoko Tagawa, MD; are from the Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center, Torrance, CA.
Disclosure: Dr. Chlebowski is a consultant for AstraZeneca, Novartis, Amgen, Genentech Genomic Health, and Novo Nordisk, and serves on the speaker’s bureaus for Novartis and Genentech.
Funding/Support: The WHI program is reported by the National Heart, Lung and Blood Institute, National Institutes of Health, Department of Health and Human Services through contracts N01WH22110, 24152, 32100-2, 32105-6, 32108-9, 32111-13, 32115, 32118-32119, 32122, 42107-26, 42129-32, and 44221.
Author Contributions: Dr. Chlebowski wrote the initial draft of the report and takes full responsibility for the report. Drs. Prabhaker and Tagawa provided critical revision of the manuscript for important intellectual content.
Acknowledgment: We thank the Women’s Health Initiative investigators, staff, and the trial participants for their outstanding dedication and commitment.
Women’s Health Initiative Investigators
Clinical Coordinating Center: (Fred Hutchinson Cancer Research Center, Seattle, WA); Garnet Anderson, Ross Prentice, Andrea LaCroix, and Charles Kooperberg
Investigators and Academic Centers: (Brigham and Women’s Hospital, Harvard Medical School, Boston, MA) JoAnn E. Manson; (Med Star Health Research Institute/Howard University, Washington, DC) Barbara V. Howard; (Stanford Prevention Research Center, Stanford, CA) Marcia L. Stefanick; (The Ohio State University, Columbus, OH) Rebecca Jackson; (University of Arizona, Tucson/Phoenix, AZ) Cynthia A. Thompson; (University at Buffalo, Buffalo, NY) Jean Wactawski-Wende; (University of Florida, Gainesville/Jacksonville, FL) Marian Limacher; (University of Iowa, Iowa City/Davenport, IA) Robert Wallace; (University of Pittsburgh, Pittsburgh, PA) Lewis Kuller; (Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center, Torrance, CA) Rowan T. Chlebowski; (Wake Forest University School of Medicine, Winston-Salem, NC) Sally Shumaker.
Women’s Health Initiative Memory Study: (Wake Forest University School of Medicine, Winston Salem, NC) Sally Shumaker.
Additional information: A full list of all the investigators who have contributed to Women’s Health Initiative science appears at https://www.whi.org/researchers/Documents%20%20Write% 20a%20Paper/WHI%20Investigator%20Long%20List.pdf.
The WHI program results in this review were funded by the National Heart, Lung, and Blood Institute at the National Institutes of Health, US Department of Health and Human Services.
References
- Visvanathan K, Chlebowski RT, Hurley P, et al. AmericanSociety of Clinical Oncology 2008 clinical practice guideline update on the use of pharmacologic interventions including tamoxifen, raloxifene, and aromatase inhibitor for breast cancer risk reduction. J Clin Oncol. 2009;27(19):3235-3258.
- National Comprehensive Cancer Network website. www.nccn. org. Accessed November 22, 2015.
- Nelson HD, Smith B, Griffin JC, Fu R. Use of medications to reduce risk for primary breast cancer: a systematic review for the US Preventive Services Task Force. Ann Intern Med. 2013;158(8):604-614.
- Waters EA, McNeel TS, Stevens WM, Freedman AN. Use of tamoxifen and raloxifene for breast cancer chemoprevention in 2010. Breast Cancer Res Treat. 2012;134(2):875-880. doi: 10.1007/ s10549-012-2089-2.
- Fisher B, Costantino JP, Wickerham DL, et al. Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel Project P-1 Study. J Natl Cancer Inst. 1998;90(18):1371-1388.
- Cuzick J, Forbes J, Edwards R, et al; IBIS investigators. First results from the International Breast Cancer Intervention Study (IBIS-I): a randomised prevention trial. Lancet. 2002;360(9336):817-824.
- Powles T, Eeles R, Ashley S, et al. Interim analysis of the incidence of breast cancer in the Royal Marsden Hospital tamoxifen randomised chemoprevention trial. Lancet. 1998;352(9122):98-101.
- Veronesi U, Maisonneuve P, Costa A, et al. Prevention of breast cancer with tamoxifen: preliminary findings from the Italian randomised trial among hysterectomised women. Italian Tamoxifen Prevention Study. Lancet. 1998;352(9122):93-97.
- Cuzick J, Sestak I, Bonanni B, et al; SERM Chemoprevention of Breast Cancer Overview Group. Selective oestrogen receptor modulators in prevention of breast cancer: an updated meta-analysis of individual participant data. Lancet. 2013;381(9880):1827-1834. doi: 10.1016/S0140-6736(13)60140- 3.
- Barrett-Connor E, Mosca L, Collins P, et al. Effects of raloxifene on cardiovascular events and breast cancer in postmenopausal women. N Engl J Med. 2006;355(2):125-137.
- Cauley JA, Norton L, Lippman ME, et al. Continued breast cancer risk reduction in postmenopausal women treated with raloxifene: 4-year results from the MORE trial. Multiple outcomes of raloxifene evaluation. Breast Cancer Res Treat. 2001;65(2):125-134. Erratum: Breast Cancer Res Treat. 2001;67(2):191.
- Martino S, Cauley JA, Barrett-Connor E, et al; CORE Investigators. Continuing outcomes relevant to Evista; breast cancer incidence in postmenopausal osteoporotic women in a randomized trial of raloxifene. J Natl Cancer Inst. 2004;96(23):1751-1761.
- Vogel VG, Costantino JP, Wickerham DL, et al; National Surgical Adjuvant Breast and Bowel Project (NSABP). Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes: the NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 Trial. JAMA. 2006;295(23):2727-2741.
- Cummings SR, Ensrud K, Delmass PD, et al. Lasofoxifene in postmenopausal women with osteoporosis. N Engl J Med. 2010;362(8):686-96. doi: 10.1056/NEJMoa0808692. Erratum: N Engl J Med. 2011;364(3):290.
- Cummings SR, McClung M, Reginster JY, et al. Arzoxifene for prevention of fractures and invasive breast cancer in postmenopausal women. J Bone Miner Res. 2011;26(2):397-404. doi: 10.1002/jbmr.191.
- Schmidt C. Third-generation SERMs may face uphill battle. J Natl Cancer Inst. 2010;102(22):1690-1692. doi: 10.1093/jnci/djq477.
- Cuzick J, Sestak I, Cawthorn S, et al; IBIS-1 Investigators. Tamoxifen for prevention of breast cancer: extended long-term follow-up of the IBIS-I breast cancer prevention trial. Lancet Oncol 2015;16(1):67-75. doi: 10.1016/S1470-2045(14)71171-4.
- Chlebowski RT. IBIS-1 tamoxifen prevention update: Maturity brings questions. Lancet Oncol. 2015;16(1):7-9. doi: 10.1016/S1470-2045(14)71184-2.
- Wickerham DL, Cecchini RS, Vogel VG, et al. Final updated of the NRG Oncology/NSABP Protocol P-2: study of tamoxifen and raloxifene (STAR) in preventing breast cancer. J Clin Oncol. 2015;33(suppl; abstr 1500).
- Early Breast Cancer Trialists’ Collaborative Group. Tamoxifen for early breast cancer: an overview of the randomised trials. Lancet. 1998;351(9114):1451-1467.
- Dowsett M, Forbes JF, Bradley R, et al; Early Breast Cancer Trialists’ Collaborative Group. Aromatase inhibitors versus tamoxifen in early breast cancer: patient-level meta-analysis of the randomized trials. Lancet. 2015;386(10001):1341-1352. doi: 10.1016/S0140-6736(15)61074-1.
- Goss PE, Ingle JN, Ales-Martin LJ, et al; NCIC CTG MAP.3 Study Investigators. Exemestane for breast-cancer prevention in postmenopausal women. N Engl J Med. 2011;364(25):2381-2391. doi: 10.1056/NEJMoa1103507.
- Cuzick J, Sestak I, Forbes JF, et al; IBIS-II Investigators. Anastrozole for prevention of breast cancer in high-risk postmenopausal women (IBIS-II) an international, double-blind, randomised placebo-controlled trial. Lancet. 2014;383(9922):1041-1048. doi:10.1016/S0140-6736(13)62292-8.
- Maunsell E, Goss PE, Chlebowski RT, et al. Quality of life in MAP.3: a randomized, placebo-controlled trial evaluating exemestane for prevention of breast cancer. J Clin Oncol. 2014;32(14):1427-1436. doi: 10.1200/JCO.2013.51.2483.
- Chlebowski RT, Kim J, Haque R. Adherence to endocrine therapy in breast cancer adjuvant and prevention settings. Cancer Prev Res (Phila). 2014;7(4):378-387. doi: 10.1158/1940-6207. CAPR-13-0389.
- Chlebowski RT, Anderson GL. Changing concepts: menopausal hormone therapy and breast cancer. J Natl Cancer Inst. 2012;104(7):517-527. doi: 10.1093/jnci/djs014.
- Chlebowski RT, Rohan TE, Manson JE, et al. Breast cancer after use of estrogen plus progestin and estrogen alone: analyses of data from two Women’s Health Initiative (WHI) randomized clinical trials. JAMA Oncol. 2015;1(3):296-305. doi: 10.1001/ jamaoncol.2015.0494.
- Rossouw JE, Anderson GL, Prentice RL, et al; Writing Group for the Women’s Health Initiative Investigators. Risk and benefits of estrogen plus progestin in healthy postmenopausal women. Principal results from the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288(3):321-333.
- Chlebowski RT, Hendrix SL, Langer RD, et al; WHI Investigators. Estrogen plus progestin influence on breast cancer and mammography in healthy postmenopausal women: the Women’s Health Initiative randomized trial. JAMA. 2003;289(24):3243-3253
- Chlebowski RT, Anderson G, Pettinger M, et al. Estrogen plus progestin and breast cancer detection with mammography and breast biopsy. JAMA. 2008;168(4):382-391.
- Chlebowski RT, Anderson GL, Gass M, et al. Influence of estrogen plus progestin on breast cancer incidence and morality. JAMA. 2010:304(15):1684-1692.
- Stefanick ML, Anderson GL, Margolis KL, et al. Effects of conjugated equine estrogens on breast cancer and mammography in postmenopausal women with hysterectomy: the Women’s Health Initiative randomized trial. JAMA. 2006;242(14):1048-1063.
- Anderson GL, Chlebowski RT, Aragaki A, et al. Oestrogen and breast cancer incidence and mortality in postmenopausal women with hysterectomy extended follow-up of the Women’s Health Initiative randomized trial. Lancet Oncol. 2012;13(5):476-486. doi: 10.1016/S1470-2045(12)70075-X.
- Hersh AL, Stefanick ML, Stafford RS. National use of menopausal hormone therapy: annual trends and response to recent evidence. JAMA. 2004;29(1):47-53.
- Zbuk K, Anand SS. Declining incidence of breast cancer after decreased use of hormone-replacement therapy: magnitude and time lags in different countries. J Epidemiol Community Health. 2012; 66(1):1-7. doi: 10.1136/jech.2008.083774.
- Clarke CA, Glaser SL, Uratsu CS, Selby JV, Kushi LH, Herrinton LJ. Recent declines in hormone therapy utilization and breast cancer incidence: clinical and population-based evidence. J Clin Oncol. 2006;24(33):e49-e50.
- Ravdin PM, Cronin KA, Howlander N, et al. The decrease in breast cancer incidence in 2003 in the United States. N Engl J Med. 2007;356(16):1670-1674.
- Chlebowski RT, Kuller L, Prentice RL, et al; WHI Investigators. Breast cancer after estrogen plus progestin use in postmenopausal women. N Engl J Med. 2009;360(6):573-587. doi: 10.1056/NEJMoa0807684.
- Manson JE, Chlebowski RT, Stefanick ML, et al. Menopausal hormone therapy and health outcomes during the intervention and extended poststopping phases of the Women’s Health Initiative randomized trials. JAMA. 2013; 310(13):1353-1368. doi: 10.1001/jama.2013.278040.
- Joshi PA, Goodwin PJ, Khokha R. Progesterone exposure and breast cancer risk: understanding the biological roots. JAMA Oncol. 2015;1(3):283-285. doi: 10.1001/jamaoncol.2015.0512.
- Chlebowski RT, Anderson GL, Aragaki AK, Prentice RL. Breast cancer and menopausal hormone therapy by race/ ethnicity and body mass index. J Natl Cancer Inst. 2015;108(2). pii:djv327. doi:10.1002.cncr.29332.
- Ellis MJ, Gao F, Jeffe DD, et al. Lower-dose vs high-dose oral estradiol therapy of hormone receptor positive advanced breast cancer. JAMA. 2009;302(7):774-780. doi: 10.1001/ jama.2009.1204.
- Santen RJ, Song Y, Yue W, Wang JP, Heitjan DF. Effects of menopausal hormonal therapy on occult breast tumors. J Steroid Biochem Mol Biol. 2013;137:150-156. doi: 10.1016/j.jsbmb.2013.05.008.
- Goodwin PJ, Stambolic V. Obesity and insulin resistance in breast cancer – chemoprevention strategies with a focus on metformin. Breast. 2011;20(suppl 3):S31-S35. doi: 10.1016/ S0960-9776(11)70291-0.
- Chlebowski RT, McTiernan A, Wactawski-Wende J, et al. Diabetes, metformin and breast cancer in postmenopausal women J Clin Oncol. 2012;30(23):2844-2852 doi: 10.1200/ JCO.2011.39.7505.
- Col N, Ochs L, Springmann V, Aragaki AK, Chlebowski RT. Metformin and breast cancer risk: a meta-analysis and critical literature review [published online July 31, 2012]. Breast Cancer Res Treat. 2012;135(3):639-646.
- Chlebowski RT. Current concepts in breast cancer chemoprevention [published online March 10, 2014]. Pol Arch Med Wewn. 2014; 124(4):191-199.
- Beral V, Reeves G, Bull D, Green J; Million Women Study Collaborators. Breast cancer risk in relation to the interval between menopause and starting hormone therapy. J Natl Cancer Inst 2011 Feb 16,103(4):296-305.
- Visvanathan K, Hurley P, Bantug E, et al. Use of pharmacologic interventions for breast cancer risk reduction: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol 2013 Aug 10;31(23):2942-62.
- Jordan VC, Ford LG. Paradoxical clinical effect of estrogen on breast cancer risk: a “new” biology of estrogen-induced apoptosis. Cancer Prev Res (Phila), 2011;4 (5): 633-637.





